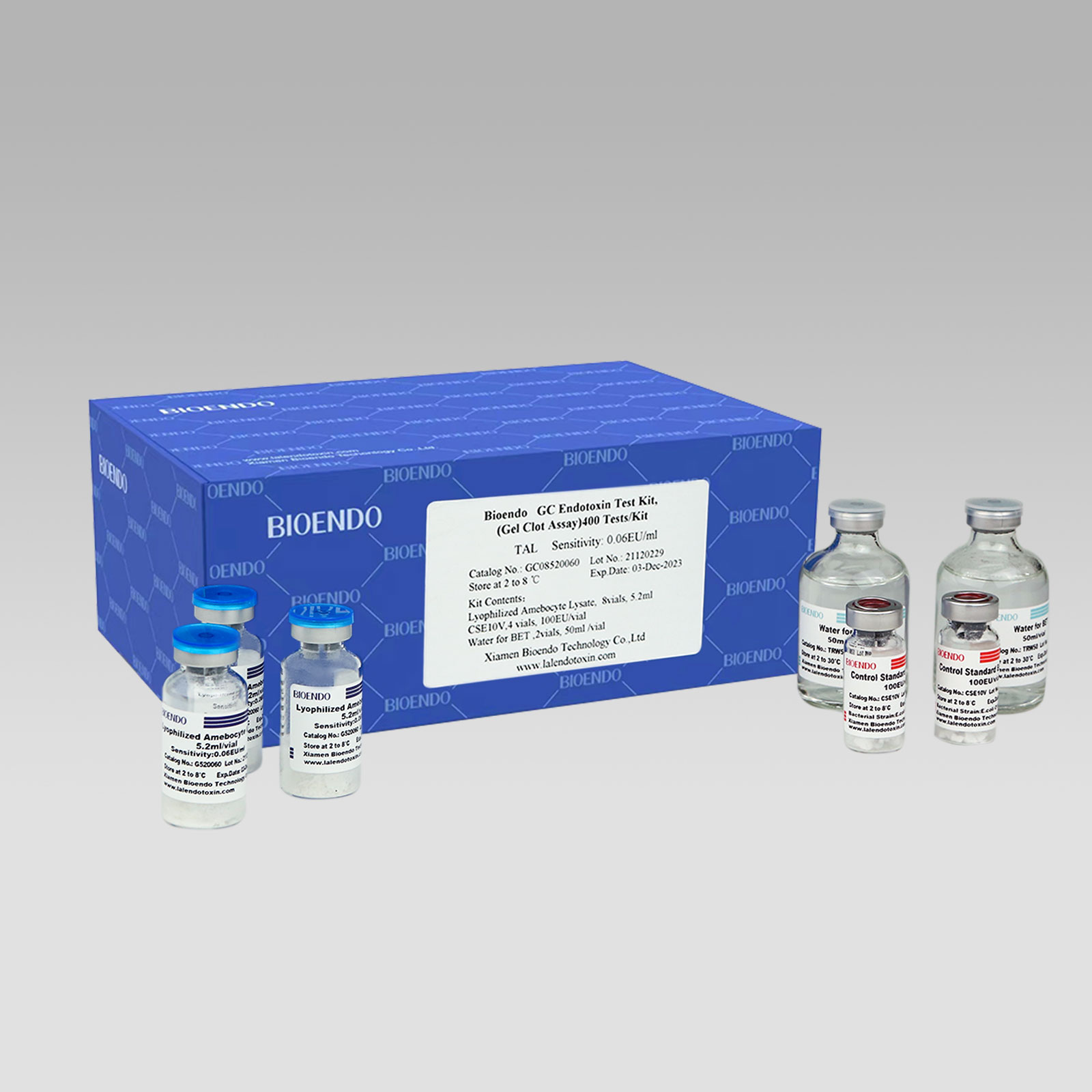
Chinese wholesale LAL test kit - Compact Modular Dry Heat Incubator – Bioendo
Chinese wholesale LAL test kit - Compact Modular Dry Heat Incubator – Bioendo Detail:
Dry Heat Incubator
1. Product description:
Dry Heat Incubator TAL-M2 is amicroprocessor-controlled device, high precision of temperature control, samplepreparation parallelism, as an alternative to the traditional water bathdevice. It is recommended to use in the gel clot TAL endotoxin assay. And it can be widely used in various other application includes the pharmaceutical, chemical, food safety, environment, quality inspection. TAL-M2 includes 2 heating modules. TAL- M2 A Dry bath incubator Can achieve two independent heating modules, thermostats, unique combo designed to meet the needs of more experimental.
2. Product features
1, LCD display, simple interface.
2, high heating speed, uniformheating, accurate temperature control, high stability, low energy consumptionand no noise.
3, Built in temperature calibrationfunction, automatic fault detection and buzzer alarm function.
4, Built in over-temperatureprotection device, safe and reliable use, prolong the service life of themachine.
5, product design, compact and tight,small occupied space, freer and easier.
6, Various blocks for convenientreplacement, cleaning and disinfection.
7,It Can achieve two independentheating modules, thermostats, unique combo designed to meet the needs of moreexperimental.
dry bath double control, dry incubator doublemodule, limulus polyphemus blue blood, tal kinetic chromogenic method, talendotoxin kit
| Product Name & Descriptions |
TAL-M2 Dry heat incubator (single heating module); One modular total 60 holes for mass test in pharmaceuticals application. |
|
| Temperature control Range | Ambient+5℃ ~ 150℃ | |
| Temperature setting range | Ambient+5℃ ~ 150℃ | |
| Temperature rising time | ≤ 30min (from 20℃ rise to 150℃), rise to 37deg C in about 60s. | |
| Temperature Stability @100~150℃ | ≤±1℃ | |
| Temperature Stability @40~100℃ | ≤±0.5℃ | |
| Temperature Uniformity @40℃ | ±0.3℃ | |
| Modular temperature uniformity | ±0.5℃ | |
| Displayed Temperature Precision | 0.1℃ | |
| Temperature Control time | 99h59min | |
| Fuse protector | 250v, 3A/6A, Φ5×20 | |
| Highest Temperature | 150℃ | |
| Power supply | AC220V/AC110V, 50/60Hz, 400W | |
| Dimension | D260*W220*H95mm | |
Product detail pictures:



Related Product Guide:
We pursue the administration tenet of "Quality is remarkable, Services is supreme, Status is first", and will sincerely create and share success with all customers for Chinese wholesale LAL test kit - Compact Modular Dry Heat Incubator – Bioendo , The product will supply to all over the world, such as: Greece, Sydney, Bandung, Our company considers that selling is not only to gain profit but also popularize the culture of our company to the world. So we are working hard to give you the wholehearted service and willing to give you the most competitive price in the market
The company has a good reputation in this industry, and finally it tured out that choose them is a good choice.