Factory wholesale Kinetic Turbidimetric LAL Assay - Kinetic Turbidimetric Amebocyte Lysate Vial – Bioendo
Factory wholesale Kinetic Turbidimetric LAL Assay - Kinetic Turbidimetric Amebocyte Lysate Vial – Bioendo Detail:
Kinetic Turbidimetric Amebocyte Lysate Vial
1. Product Introduction
Kinetic Turbidimetric Amebocyte Lysate Vial is developed based on the principle that the time needed to reach a certain absorbance increase (onset OD), i.e. onset time, is negatively correlated with the endotoxin concentration. Sensitivity could reach 0.005EU/ml, and the detection could reach four orders of magnitude. It is specially suitable for pharmaceuticals industry to monitor endotoxin concentration.
2. Product Parameter:
Assay range:0.005-50EU/ml; 0.01 – 10EU/ml
3. Product Application
End-product endotoxin (pyrogen) qualification, Water for injection endotoxin assay, raw material endotoxin testing or endotoxin level monitoring during manufacturing process for pharmaceutical companies or medical devices manufacturers.
Note:
Lyophilized Amebocyte Lysate reagent manufactured by Bioendo is made from amebocyte lysate from the horseshoe crab (Tachypleus tridentatus).
|
Catalog No. |
ml/vial |
Tests/Vial |
Vials/Pack |
Sensitivity EU/ml |
|
KT17 |
1.7 |
16 |
10 |
0.01-10EU/ml |
|
KT17S |
1.7 |
16 |
10 |
0.005-5EU/ml, 0.01-10EU/ml |
|
KT52 |
5.2 |
50 |
10 |
0.01-10EU/ml |
|
KT52S |
5.2 |
50 |
10 |
0.005-5EU/ml, 0.01-10EU/ml |
The Lyophilized Amebocyte Lysate reagent sensitivity and the Control Standard Endotoxin potency are assayedagainst USP Reference Standard Endotoxin. The Lyophilized Amebocyte Lysate reagent kits come withproduct instruction, Certificate of Analysis.
Product detail pictures:
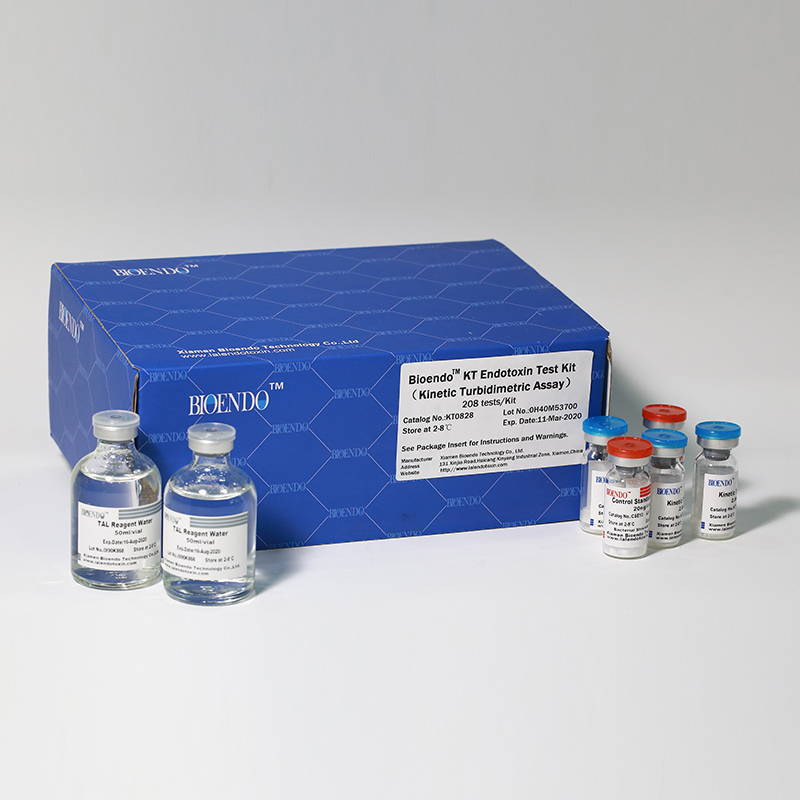
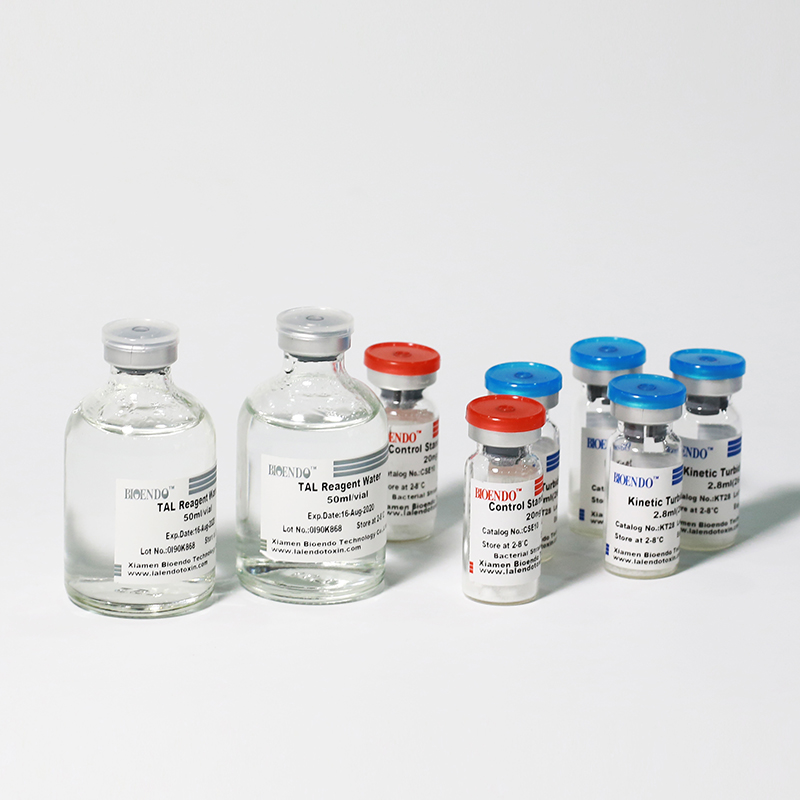

Related Product Guide:
Our organization has been focusing on brand strategy. Customers' gratification is our greatest advertising. We also source OEM provider for Factory wholesale Kinetic Turbidimetric LAL Assay - Kinetic Turbidimetric Amebocyte Lysate Vial – Bioendo , The product will supply to all over the world, such as: Croatia, Swaziland, Leicester, Insisting on the high quality generation line management and customers expert assistance, we now have designed our resolution to offer our buyers using the to start with amount getting and just after services practical experience. Maintaining the prevailing friendly relations with our buyers, we however innovate our solution lists all of the time to satisfy the brand new demands and adhere to the most up-to-date development of the market in Malta. We are ready to face the worries and make the improve to understand all the possibilities in international trade.
The factory can meet continuously developing economic and market needs, so that their products are widely recognized and trusted, and that's why we chose this company.







