Factory wholesale TAL Reagent Water - Single-Channel Mechanical Pipettor – Bioendo
Factory wholesale TAL Reagent Water - Single-Channel Mechanical Pipettor – Bioendo Detail:
Single-Channel Mechanical Pipettor
1. Product Information
Single channel mechanical pipette is ideal tool to support endotoxin detection with Lyophilized Amebocyte Lysate which covers gel-clot technique, kinetic turbidimetric technique, kinetic chromogenic technique, and end-point chromogenic technique. All pipettors are produced by following ISO8655 – 2:2002. The quality control involves gravimetric testing of each pipette with distilled water at 22℃.
2. Product Features:
- Light weight, economic, low force design
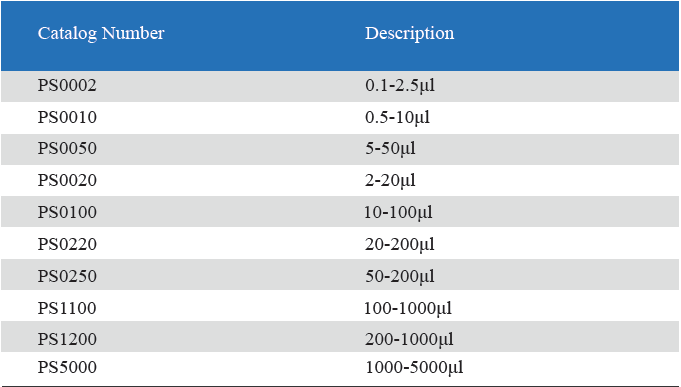
- Measuring volume range from 0.1μL to 5mL
- Easy to calibrate and maintain with tool supplied
- Design helps to avoid repetitive strain injuries
- Calibrated in accordance with ISO8655. Each pipettor supplied with individual test certificate
- The low part is available for autoclaving
Product detail pictures:

Related Product Guide:
We retain improving upon and perfecting our items and repair. At the same time, we perform actively to do research and progress for Factory wholesale TAL Reagent Water - Single-Channel Mechanical Pipettor – Bioendo , The product will supply to all over the world, such as: Turkmenistan, Curacao, Cape Town, we are now looking forward to even greater cooperation with overseas customers based on mutual benefits. We will work wholeheartedly to improve our products and services. We also promise to work jointly with business partners to elevate our cooperation to a higher level and share success together. Warmly welcome you to visit our factory sincerely.
The factory workers have a good team spirit, so we received high quality products fast, in addition, the price is also appropriate, this is a very good and reliable Chinese manufacturers.






