Manufactur standard Endotossine LAL Test - Mini Dry Heat Incubator – Bioendo
Manufactur standard Endotossine LAL Test - Mini Dry Heat Incubator – Bioendo Detail:
Dry heat incubator single module
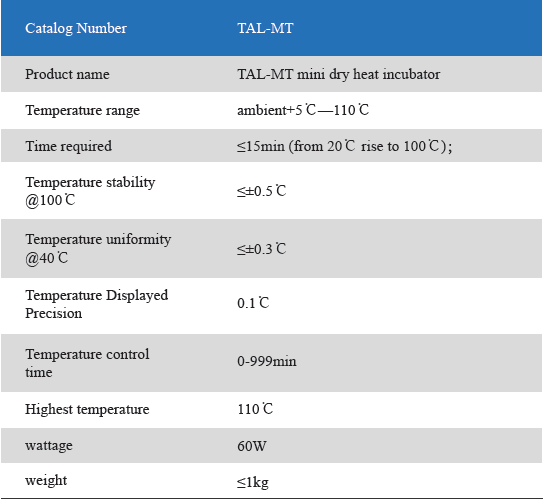
1. Product information
The Mini Dry Heat Incubator is a micro-processor controlled heating block with semi conductor heating technology.It adapts onboard use, smart, light and convenient for movement, suit for any kind of occasions. Especially good for the incubation of the gel clot LAL assay, LAL chromogenic endpoint assay incubation.
2. Product features
1. Unique designed. Smart and light, convenient movement, suit for various occasions.
2. LCD simultaneously display setting and actual time and temperature.Temperature calibration function.
3. Automatic fault detection function with buzzer alarm.
4. 24V DC input power, built-in over-temperature protection device.
5. Various of blocks for optional choice. Convenient for replacement. Easy cleaning and disinfection.
Product detail pictures:



Related Product Guide:
Dedicated to strict quality management and thoughtful client services, our experienced staff customers are generally available to discuss your demands and guarantee full client pleasure for Manufactur standard Endotossine LAL Test - Mini Dry Heat Incubator – Bioendo , The product will supply to all over the world, such as: Sheffield, Bahamas, Brisbane, We have constructed strong and long co-operation relationship with an enormous quantity of companies within this business in Kenya and overseas. Immediate and specialist after-sale service supplied by our consultant group has happy our buyers. Detailed Info and parameters from the merchandise will probably be sent to you for any thorough acknowledge. Free samples may be delivered and company check out to our corporation. n Kenya for negotiation is constantly welcome. Hope to get inquiries type you and construct a long-term co-operation partnership.
The company comply with the contract strict, a very reputable manufacturers, worthy a long-term cooperation.







