Manufacturer of Dry Heat Sterilization Validation – Mini Dry Heat Incubator – Bioendo
Manufacturer of Dry Heat Sterilization Validation – Mini Dry Heat Incubator – Bioendo Detail:
Dry heat incubator single module
1. Product information
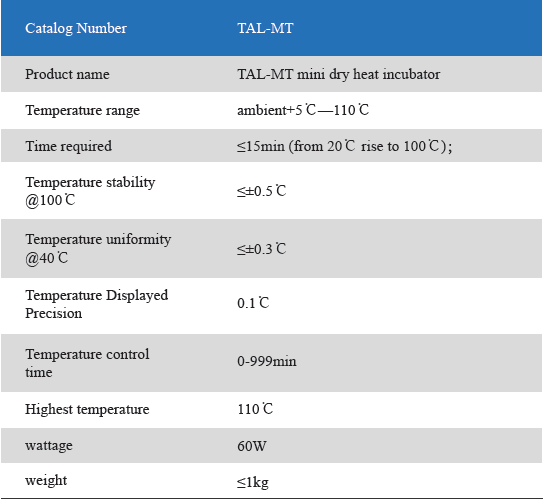
The Mini Dry Heat Incubator is a micro-processor controlled heating block with semi conductor heating technology.It adapts onboard use, smart, light and convenient for movement, suit for any kind of occasions. Especially good for the incubation of the gel clot LAL assay, LAL chromogenic endpoint assay incubation.
2. Product features
1. Unique designed. Smart and light, convenient movement, suit for various occasions.
2. LCD simultaneously display setting and actual time and temperature.Temperature calibration function.
3. Automatic fault detection function with buzzer alarm.
4. 24V DC input power, built-in over-temperature protection device.
5. Various of blocks for optional choice. Convenient for replacement. Easy cleaning and disinfection.
Product detail pictures:



Related Product Guide:
Our products are broadly identified and trustworthy by people and may meet continually modifying financial and social requires of Manufacturer of Dry Heat Sterilization Validation – Mini Dry Heat Incubator – Bioendo , The product will supply to all over the world, such as: UAE, Poland, French, We hope we can establish long-term cooperation with all of the customers. And hope we can improve competitiveness and achieve the win-win situation together with the customers. We sincerely welcome the customers from all over the world to contact us for anything you need!
Problems can be quickly and effectively resolved, it is worth to be trust and working together.