OEM Manufacturer Endotoxin Assay Kit - Control Standard Endotoxin (CSE) – Bioendo
OEM Manufacturer Endotoxin Assay Kit - Control Standard Endotoxin (CSE) – Bioendo Detail:
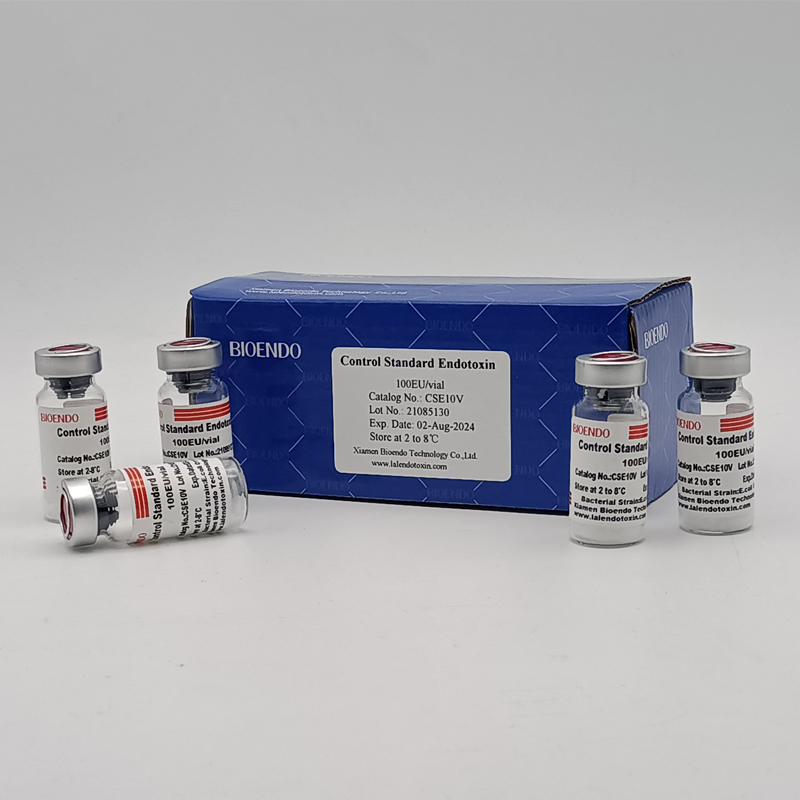
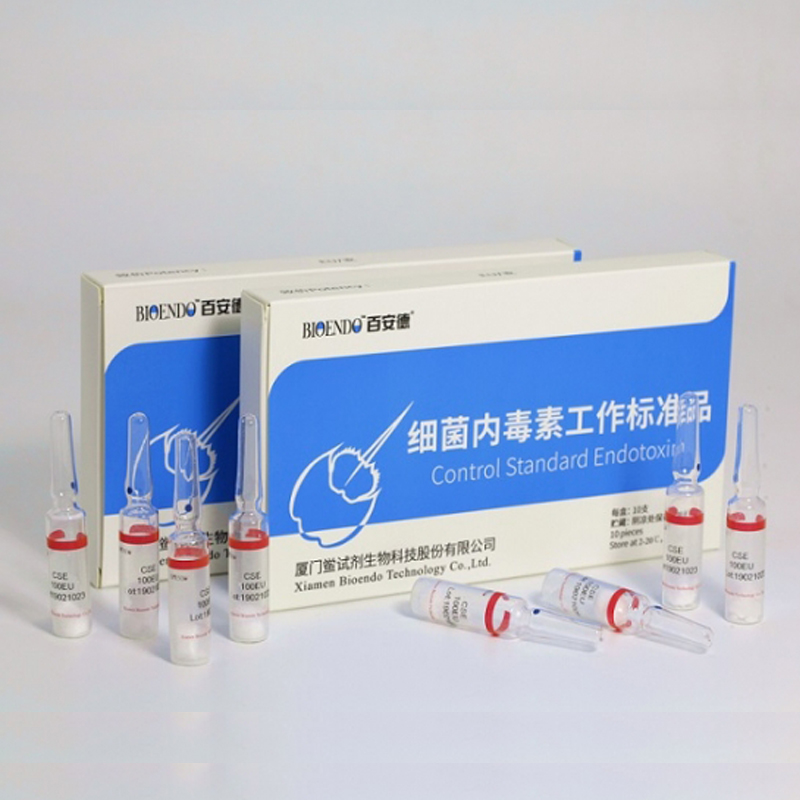
Control Standard Endotoxin (CSE)
1. Product Information
Control Standard Endotoxin (CSE) is extracted from E.coli O111:B4. CSE is an economic alternative to Reference Standard Endotoxin (RSE) in constructing standard curves, validating product and preparing controls in Lyophilized Amebocyte Lysate test. The labeled potency of CSE endotoxinE.coli standard is referenced against RSE. The Control Standard Endotoxin could be used with gel clot assay, kinetic turbidimetric assay or kinetic chromogenic assay as the endotoxin testing standards. The Certificate of Analysis will show the matched Lyophilized Amebocyte Lysate reagent lots.
2. Product Parameter
| Catalog Number | Potency (EU/vial) | Package |
| CSE10V | 100 to 999 EU | seal in glass vial, 10vials/pack |
| CSE100V | 1 to 199 EU | seal in glass vial, 10vials/pack |
| CSE10A | 1 to 99 EU | seal in glass ampoule, 10vials/pack |
3. Product Feature and Application
Bioendo CSE was labeled by the potency and matched to Lyophilized Amebocyte Lysate reagent lots. Users do not need to do the CSE/RSE ratio assay. Low potency control standard endotoxin is available to avoid lots of steps of dilution to provide convenience for end users.
Product Condition:
Control Standard Endotoxin (CSE), extracted from E.coli O111:B4, is an economic alternative to Reference Standard Endotoxin (RSE) in constructing standard curves, validating product and preparing controls in endotoxin test. The potency of CSE is referenced against USP Reference Standard Endotoxin, and labeled in the Certificate of Analysis.
Endotoxin test assay: Lysate reagent and CSE lot number have to be matched.
Pyrogen free tip box
Endotoxin free tubes
Product detail pictures:


Related Product Guide:
We will make every effort to be outstanding and perfect, and accelerate our steps for standing in the rank of international top-grade and high-tech enterprises for OEM Manufacturer Endotoxin Assay Kit - Control Standard Endotoxin (CSE) – Bioendo , The product will supply to all over the world, such as: Rome, Uganda, Guinea, In order to carry out our goal of "customer first and mutual benefit" in the cooperation, we establish a professional engineering team and a sales team to supply the best service to satisfy our customers' requirements. Welcome you to cooperate with us and join us. We are your best choice.
This supplier's raw material quality is stable and reliable, has always been in accordance with the requirements of our company to provide the goods that quality meet our requirements.







