OEM/ODM Manufacturer Endotoksin Testi - Single-Channel Mechanical Pipettes (Semi-sterile) – Bioendo
OEM/ODM Manufacturer Endotoksin Testi - Single-Channel Mechanical Pipettes (Semi-sterile) – Bioendo Detail:
Single-Channel Mechanical Pipettes (Semi-sterile)
1. Product Information
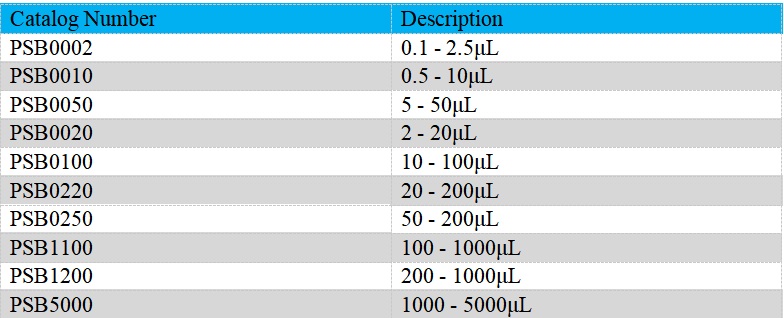
The Single-Channel Mechanical Pipettes (Semi-sterile) is adjustable, which could be used to dispense liquid precisely. Measuring volume of our Single-Channel Mechanical Pipettes (Semi-sterile) ranges from 0.1μL to 5mL. Products are produced based on ISO8655/DIN12650. It could be widely employed on endotoxin detection, etc..
2. Product Features
- Light weight, economic
- The pipettes cover volume range from 0.1μL to 5mL
- Semi-sterile
- Easy calibration and maintenance
Product detail pictures:

Related Product Guide:
We now have a specialist, efficiency workforce to deliver excellent service for our purchaser. We always follow the tenet of customer-oriented, details-focused for OEM/ODM Manufacturer Endotoksin Testi - Single-Channel Mechanical Pipettes (Semi-sterile) – Bioendo , The product will supply to all over the world, such as: Mexico, Romania, Surabaya, Our company is working by the operation principle of "integrity-based, cooperation created, people oriented, win-win cooperation". We hope we can have a friendly relationship with businessman from all over the world.
The company account manager has a wealth of industry knowledge and experience, he could provide appropriate program according our needs and speak English fluently.






