OEM/ODM Supplier Kit LAL - Single-Channel Mechanical Pipettor – Bioendo
OEM/ODM Supplier Kit LAL - Single-Channel Mechanical Pipettor – Bioendo Detail:
Single-Channel Mechanical Pipettor
1. Product Information
Single channel mechanical pipette is ideal tool to support endotoxin detection with Lyophilized Amebocyte Lysate which covers gel-clot technique, kinetic turbidimetric technique, kinetic chromogenic technique, and end-point chromogenic technique. All pipettors are produced by following ISO8655 – 2:2002. The quality control involves gravimetric testing of each pipette with distilled water at 22℃.
2. Product Features:
- Light weight, economic, low force design
- Measuring volume range from 0.1μL to 5mL
- Easy to calibrate and maintain with tool supplied
- Design helps to avoid repetitive strain injuries
- Calibrated in accordance with ISO8655. Each pipettor supplied with individual test certificate
- The low part is available for autoclaving
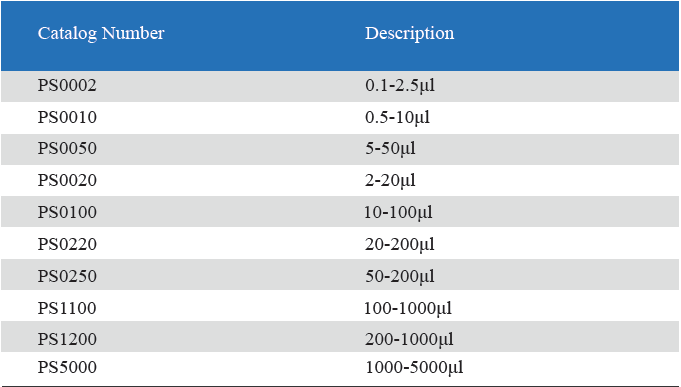
Product detail pictures:

Related Product Guide:
We've one of the most advanced generation tools, experienced and qualified engineers and workers, recognized good quality manage systems and a friendly skilled product sales workforce pre/after-sales support for OEM/ODM Supplier Kit LAL - Single-Channel Mechanical Pipettor – Bioendo , The product will supply to all over the world, such as: Panama, United States, India, Our company insists on the purpose of "takes service priority for standard, quality guarantee for the brand, do business in good faith, to provide professional, rapid, accurate and timely service for you". We welcome old and new customers to negotiate with us. We will serve you with all sincerity!
This is a very professional and honest Chinese supplier, from now on we fell in love with the Chinese manufacturing.