Rapid Delivery for Endotoxin Producing Bacteria - Control Standard Endotoxin (CSE) – Bioendo
Rapid Delivery for Endotoxin Producing Bacteria - Control Standard Endotoxin (CSE) – Bioendo Detail:
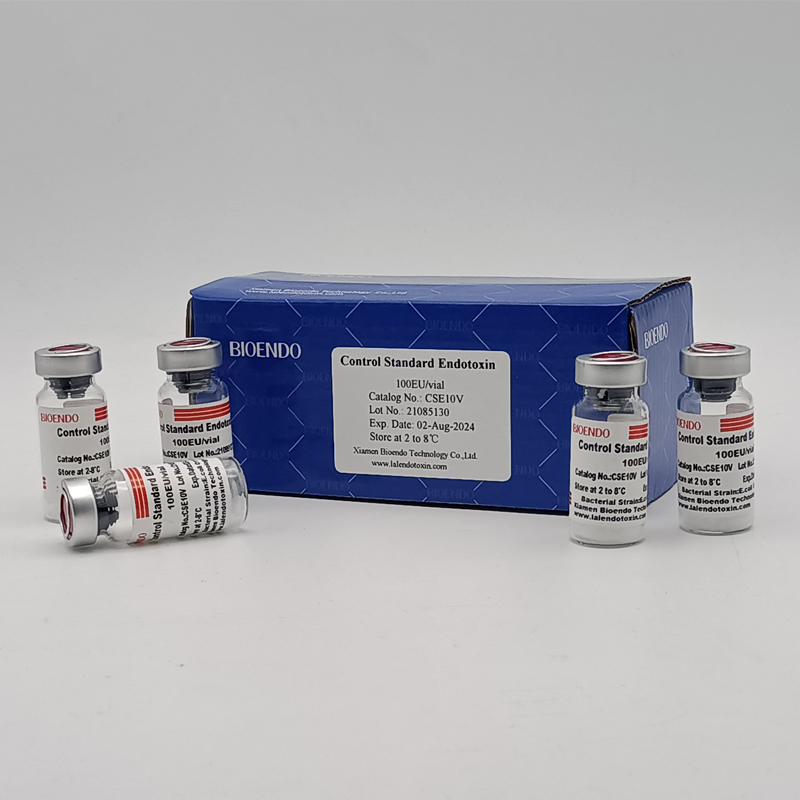
Control Standard Endotoxin (CSE)
1. Product Information
Control Standard Endotoxin (CSE) is extracted from E.coli O111:B4. CSE is an economic alternative to Reference Standard Endotoxin (RSE) in constructing standard curves, validating product and preparing controls in Lyophilized Amebocyte Lysate test. The labeled potency of CSE endotoxinE.coli standard is referenced against RSE. The Control Standard Endotoxin could be used with gel clot assay, kinetic turbidimetric assay or kinetic chromogenic assay as the endotoxin testing standards. The Certificate of Analysis will show the matched Lyophilized Amebocyte Lysate reagent lots.
2. Product Parameter
| Catalog Number | Potency (EU/vial) | Package |
| CSE10V | 100 to 999 EU | seal in glass vial, 10vials/pack |
| CSE100V | 1 to 199 EU | seal in glass vial, 10vials/pack |
| CSE10A | 1 to 99 EU | seal in glass ampoule, 10vials/pack |
3. Product Feature and Application
Bioendo CSE was labeled by the potency and matched to Lyophilized Amebocyte Lysate reagent lots. Users do not need to do the CSE/RSE ratio assay. Low potency control standard endotoxin is available to avoid lots of steps of dilution to provide convenience for end users.
Product Condition:
Control Standard Endotoxin (CSE), extracted from E.coli O111:B4, is an economic alternative to Reference Standard Endotoxin (RSE) in constructing standard curves, validating product and preparing controls in endotoxin test. The potency of CSE is referenced against USP Reference Standard Endotoxin, and labeled in the Certificate of Analysis.
Endotoxin test assay: Lysate reagent and CSE lot number have to be matched.
Pyrogen free tip box
Endotoxin free tubes
Product detail pictures:


Related Product Guide:
We insist on offering good quality generation with very good business enterprise concept, honest income as well as best and fast assistance. it will bring you not only the premium quality product or service and huge profit, but probably the most significant is usually to occupy the endless market for Rapid Delivery for Endotoxin Producing Bacteria - Control Standard Endotoxin (CSE) – Bioendo , The product will supply to all over the world, such as: Serbia, Eindhoven, Brasilia, For anyone who is keen on any of our goods right after you view our product list, please really feel absolutely free to get in touch with us for inquiries. You are able to send us emails and contact us for consultation and we shall respond to you as soon as we can. If it's easy, you may locate out our address in our web-site and come to our business for far more information of our products by your self. We are always ready to construct extended and steady co-operation relations with any possible customers in the related fields.
This is a very professional wholesaler, we always come to their company for procurement, good quality and cheap.








