Special Design for Bacterial Endotoxin Test Methods - Depyrogenated Sample Bottles ( Depyrogenated Galssware ) – Bioendo
Special Design for Bacterial Endotoxin Test Methods - Depyrogenated Sample Bottles ( Depyrogenated Galssware ) – Bioendo Detail:
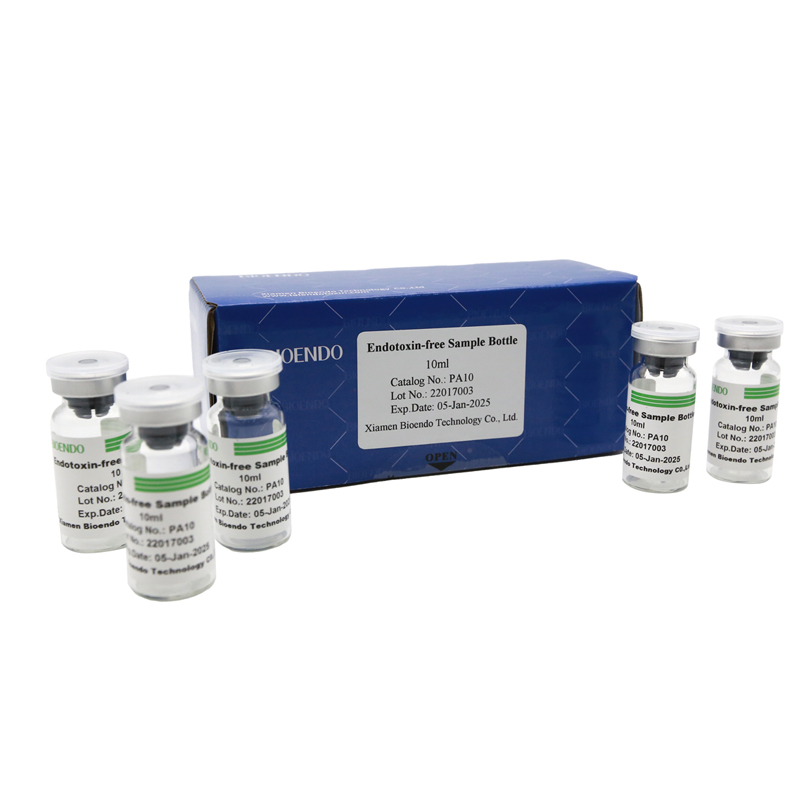
Depyrogenated Sample Bottle
1. Product information
We offer various of low endotoxin, pyrogen free accessories products,includes Water for Bacterial Endotoxins Test, pyrogen-free test tubes, pyrogen-free pipette tips, pyrogen free microplates and sample bottles for your conveniences. Amoung the sample bottle have two types, one is depyrogenated glassware and the other is depyrogenated plasticware, both endotoxin free level. High quality depyrogenated low endotoxin pyrogen free products insure the success of your experiments.
Depyrogenated (Endotoxin Free) Sample container(endotoxin free bottle, pyrogen free bottle, pyrogen free sample bottle)are glass bottles contain less than 0.005 EU/ml endotoxin. These bottles could be used to store various samples for lps endotoxin test, such as protein solution, vaccines, DNA solutions, dialysate, water for injections, etc., for endotoxin testing. Come with endotoxin free seals.
2. Product parameter
Endotoxin level < 0.005 EU/ml
3. Product features and application
For preparing and storage of the test samples.
| Catalog Number | Descriptions | Package |
| PA2 | Endotoxin free sample glass ampoule, 2ml | 10Pcs/Pack |
| PA10 | Endotoxin free sample glass vial, 10ml | 10Pcs/Pack or 110Pcs/Pack |
| PA50 | Endotoxin free sample glass vial, 50ml | 10Pcs/Pack |
| PA125 | Pyrogen free sample bottle, 125ml | 1Pcs/Pack |
| PA500 | Pyrogen free sample bottle, 500ml | 1Pcs/Pack |
Product detail pictures:





Related Product Guide:
In the past few years, our business absorbed and digested advanced technologies both equally at home and abroad. In the meantime, our company staffs a group of experts devoted to your advancement of Special Design for Bacterial Endotoxin Test Methods - Depyrogenated Sample Bottles ( Depyrogenated Galssware ) – Bioendo , The product will supply to all over the world, such as: Germany, Detroit, Ukraine, There are advanced producing & processing equipment and skilled workers to ensure the products with high quality. We have found an excellent before-sale, sale, after-sale service to ensure the customers that could rest assured to make orders. Until now our products are now moving on fast and very popular in South America, East Asia, the Middle east, Africa, etc.
Hope that the company could stick to the enterprise spirit of "Quality, Efficiency, Innovation and Integrity", it will be better and better in the future.