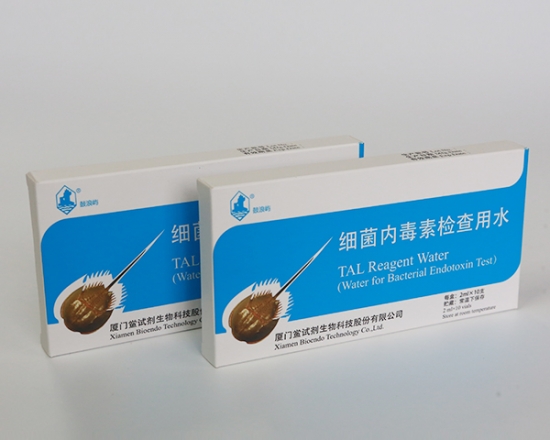
LAL Reagent Water (Water for Bacterial Endotoxins Test)
LAL Reagent Water ( Water for Bacterial Endotoxins Test )
1. Product Information
LAL Reagent Water (Water for Bacterial Endotoxins Test or BET Water or Water for BET ) is specially processed super-purified endotoxin free water is used for Endotoxin test. Its endotoxin concentration is less than 0.005 EU/ml. Various packages, such as 2ml, 10ml, 50ml, 100ml and 500ml per unit, are provided for users’ convenience. LAL Reagent Water (Water for BET) could be used to dilute the assay sample, construct the stand curve, or reconstitute the Lyophilized Amebocyte Lysate reagents.
2. Product Parameter
Endotoxin level: ≤0.005 EU/ml
Water for Bacterial Endotoxins Test is specially processed water is used for endotoxin detection. Its endotoxin concentration is less than 0.005EU/ml. We also provide water for BET with endotoxin level less than 0.001EU/ml for the assay sensitivity 0.001 to 5EU/ml kinetic chromogenic assay.
3. Product Features and Application
Endotoxin Free Water (Water for BET, LAL Reagent water, Endotoxin free water or BET water) is specially processed water intended for reconstitution of Lyophilized Amebocyte Lysate and Control Standard Endotoxin (CSE), and to dilute samples and control standards in the endotoxin assay operation.
For reconstitution of Lyophilized Amebocyte Lysate or Amebocyte Lysate, dilution of the test samples and Control Standard Endotoxin, preparation endotoxin free buffers, and construction of the standard curve. 500ml BET water main apply in extract endotoxins from medical devices.
|
Catalog No. |
Volume (ml/vial) |
Package |
|
TRW02 |
2ml in Ampoule |
In Ampoule, 10 Ampoules/Pack |
|
TRW05 |
5ml in Ampoule |
In Ampoule, 10 Ampoules/Pack |
|
TRW10 |
10ml in Ampoule |
In Ampoule, 10 Ampoules/Pack |
|
TRW50 |
50ml in Glass Vial |
In Glass Vial, 10 Vials/Pack |
|
TRW100 |
100ml in Glass Vial |
In Glass Vial, 10 Vials/Pack |
|
TRW500 |
500ml in Plastic Bottle |
1 Bottle |
The sensitivity of Lyophilized Amebocyte Lysate and potency of Control Standard Endotoxin are assayed against USP Reference Standard Endotoxin.
The Lyophilized Amebocyte Lysate reagent kits come with product instruction, Certificate of Analysis.
What is BET water (bacterial endotoxin test water)?
Endotoxin test in water that is endotoxin-free level water, it mainly use for endotoxin test assay in the operation of reconstitution and dilution.
Such as, reconstitute lyophilized lysate reagent, control standard endotoxin, and samples dilutions. also use in the sampling extraction and treatment.
Bioendo BET water (Water for BET) can be named TAL reagent water or LAL reagent water. ” LAL (lyophilized amebocyte lysate) reagent ” .
Introducing our Water for BET solution for testing endotoxin in medical devices especially for the immersing or dissolving samples. As a crucial step in ensuring the safety and reliability of medical devices, our product is designed to streamline the sampling extraction procedures by providing a larger volume of LAL reagent water. By offering a more efficient and effective solution, BET water item aims to simplify the process of endotoxin testing for medical devices, ultimately saving time and resources for our customers.
BET water series is specifically tailored to address the challenges and complexities of endotoxin testing for medical devices. With the need for a larger volume of LAL reagent water in the sampling extraction procedures, our solution is the perfect fit for laboratories and medical device manufacturers looking to enhance their testing processes. By providing a more substantial volume of LAL reagent water, our product facilitates a more thorough and comprehensive testing procedure, ensuring accurate results and peace of mind for our customers.
In addition to its practical benefits, the series also boasts a user-friendly design and seamless integration into existing testing protocols. With a focus on simplicity and efficiency, our solution can be easily incorporated into laboratory workflows and testing procedures. By streamlining the sampling extraction procedures and offering a larger volume of LAL reagent water, the water empowers our customers to enhance the quality and reliability of their endotoxin testing for medical devices. We are confident that our innovative solution will make a significant impact on the endotoxin testing process, ultimately contributing to the safety and efficacy of medical devices in the healthcare industry.