Wholesale Kinetic Turbidimetric TAL Test - Kinetic Turbidimetric Amebocyte Lysate Vial – Bioendo
Wholesale Kinetic Turbidimetric TAL Test - Kinetic Turbidimetric Amebocyte Lysate Vial – Bioendo Detail:
Kinetic Turbidimetric Amebocyte Lysate Vial
1. Product Introduction
Kinetic Turbidimetric Amebocyte Lysate Vial is developed based on the principle that the time needed to reach a certain absorbance increase (onset OD), i.e. onset time, is negatively correlated with the endotoxin concentration. Sensitivity could reach 0.005EU/ml, and the detection could reach four orders of magnitude. It is specially suitable for pharmaceuticals industry to monitor endotoxin concentration.
2. Product Parameter:
Assay range:0.005-50EU/ml; 0.01 – 10EU/ml
3. Product Application
End-product endotoxin (pyrogen) qualification, Water for injection endotoxin assay, raw material endotoxin testing or endotoxin level monitoring during manufacturing process for pharmaceutical companies or medical devices manufacturers.
Note:
Lyophilized Amebocyte Lysate reagent manufactured by Bioendo is made from amebocyte lysate from the horseshoe crab (Tachypleus tridentatus).
|
Catalog No. |
ml/vial |
Tests/Vial |
Vials/Pack |
Sensitivity EU/ml |
|
KT17 |
1.7 |
16 |
10 |
0.01-10EU/ml |
|
KT17S |
1.7 |
16 |
10 |
0.005-5EU/ml, 0.01-10EU/ml |
|
KT52 |
5.2 |
50 |
10 |
0.01-10EU/ml |
|
KT52S |
5.2 |
50 |
10 |
0.005-5EU/ml, 0.01-10EU/ml |
The Lyophilized Amebocyte Lysate reagent sensitivity and the Control Standard Endotoxin potency are assayedagainst USP Reference Standard Endotoxin. The Lyophilized Amebocyte Lysate reagent kits come withproduct instruction, Certificate of Analysis.
Product detail pictures:
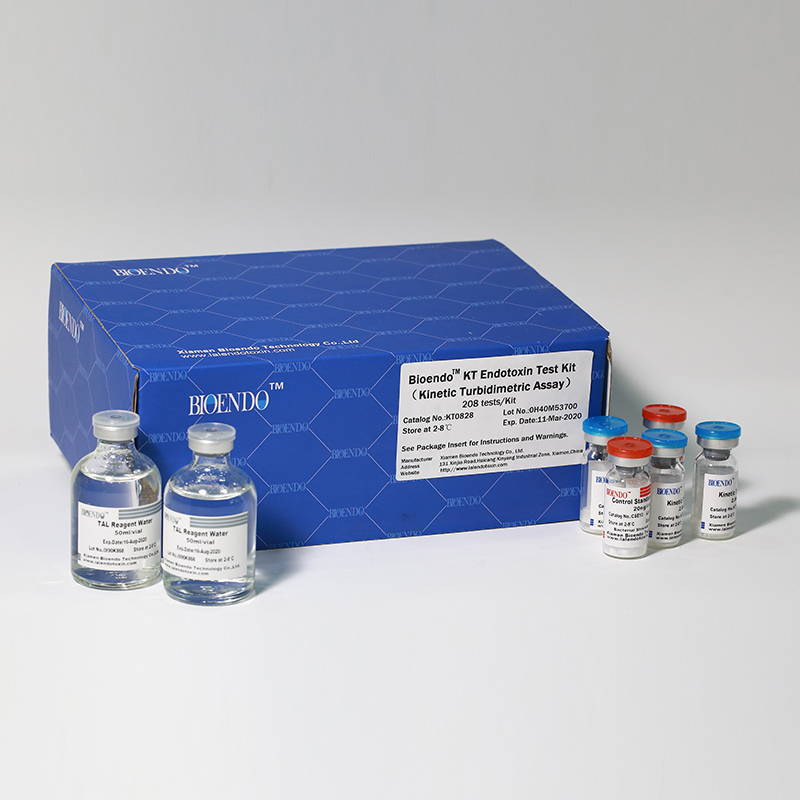
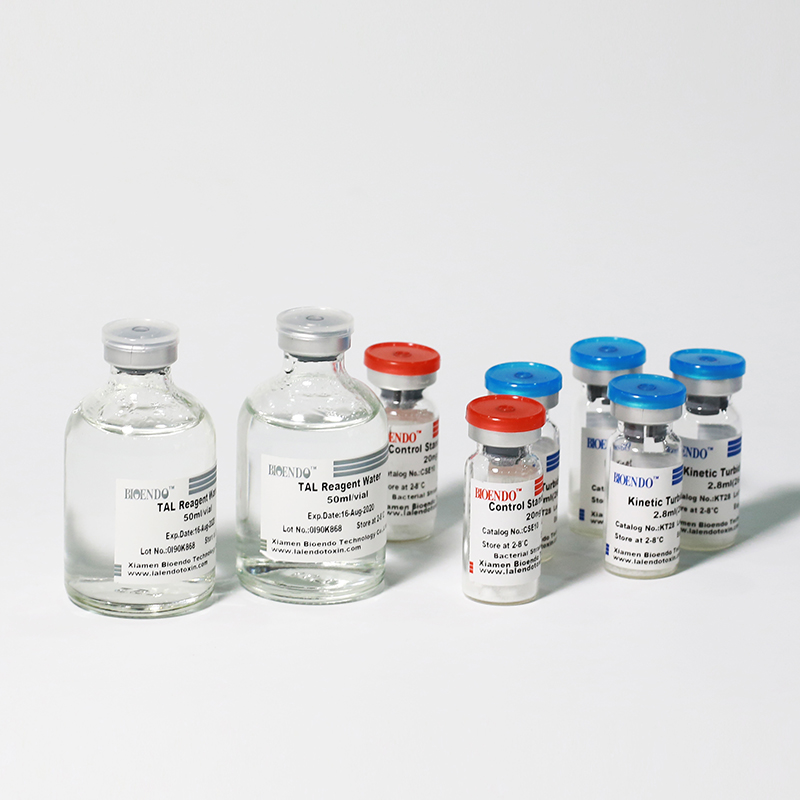

Related Product Guide:
We believe that prolonged time period partnership is really a result of top of the range, benefit added provider, prosperous knowledge and personal contact for Wholesale Kinetic Turbidimetric TAL Test - Kinetic Turbidimetric Amebocyte Lysate Vial – Bioendo , The product will supply to all over the world, such as: Bahamas, Nigeria, Luxemburg, Since always, we adhering to the "open and fair, share to get, the pursuit of excellence, and creation of value"values, adhere to the"integrity and efficient, trade-oriented, best way , best valve" business philosophy. Together with our all over the world have branches and partners to develop new business areas, maximum common values. We sincerely welcome and together we share in global resources, opening up new career together with the chapter.
Speaking of this cooperation with the Chinese manufacturer, I just want to say"well dodne", we are very satisfied.








